Diffusion and Mass Transport in Materials (Basics)+50 
Diffusion and mass transport in materials refer to the movement of atoms, ions, or molecules within solids, liquids, or gases. This process is fundamental in materials science, as it governs how substances mix, react, or change properties over time. Understanding diffusion helps explain phenomena such as alloy formation, corrosion, sintering, and phase transformations, as well as the performance of materials in various applications, including batteries, semiconductors, and structural components.
Diffusion and Mass Transport in Materials (Basics)+50 

Diffusion and mass transport in materials refer to the movement of atoms, ions, or molecules within solids, liquids, or gases. This process is fundamental in materials science, as it governs how substances mix, react, or change properties over time. Understanding diffusion helps explain phenomena such as alloy formation, corrosion, sintering, and phase transformations, as well as the performance of materials in various applications, including batteries, semiconductors, and structural components.
💡 Key Takeaways
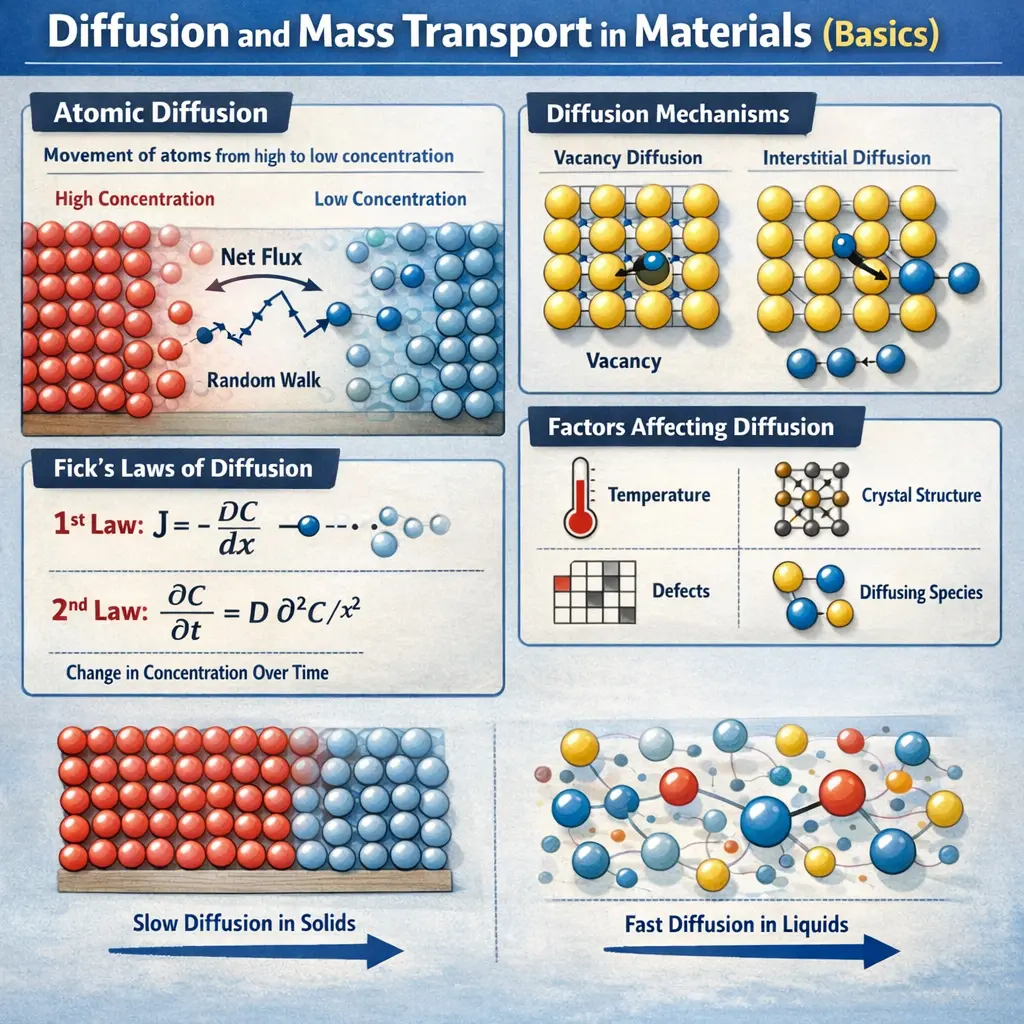
- Diffusion is the mass transport driven by concentration differences in materials.
- Fick's laws describe diffusion flux and how diffusion evolves over time (steady-state and transient diffusion).
- Primary diffusion mechanisms in solids include vacancy diffusion and interstitial diffusion, both strongly temperature dependent.
- Diffusion influences material properties and processing outcomes (e.g., alloying, phase formation, heat treatment, and corrosion).
❓ Frequently Asked Questions
What is diffusion in materials science?
The net movement of atoms or molecules from regions of high concentration to low concentration, driven by a chemical potential gradient. In solids, diffusion can occur via lattice sites, vacancies, interstitials, or along defects like grain boundaries.
What are Fick's laws and what do they mean in simple terms?
Fick's first law: diffusive flux J = -D ∇C, meaning substances move from high to low concentration. Fick's second law: ∂C/∂t = D ∇^2 C, describing how concentration changes over time with diffusion.
What factors affect diffusion rates in solids?
Temperature (higher T increases diffusion), diffusion path (lattice, grain boundaries, dislocations), crystal structure, defects, and the magnitude of the concentration gradient.
How is mass transport related to diffusion?
Mass transport is the overall movement of material; diffusion is a primary mechanism of mass transport in solids, driven by concentration gradients. In fluids, convection and electromigration can also contribute.
