British contributions to the periodic table and chemistry
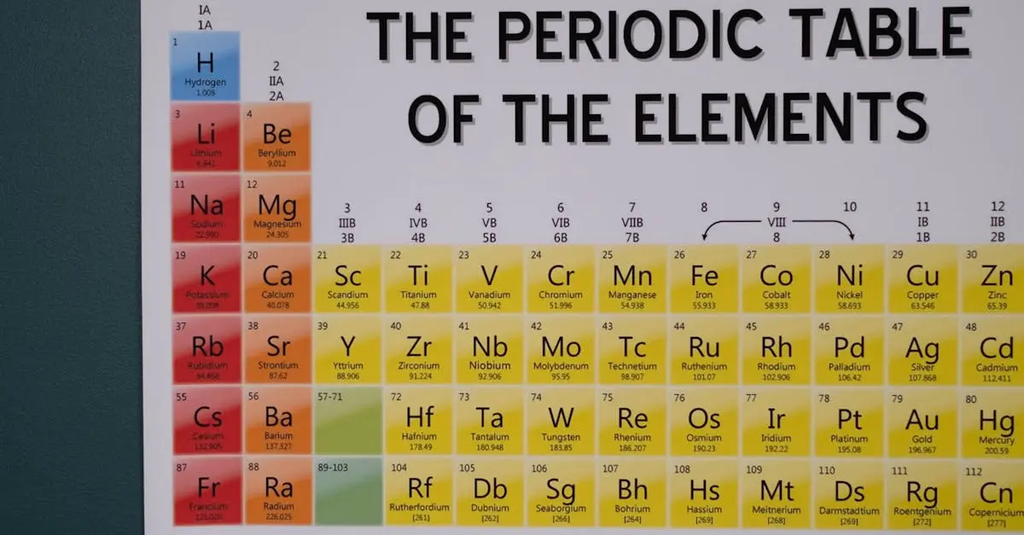
British scientists have made significant contributions to the periodic table and chemistry. John Dalton developed atomic theory, laying the groundwork for understanding chemical elements. Sir Humphry Davy discovered several elements, including sodium and potassium. Henry Moseley’s work on atomic numbers redefined the periodic table’s structure. Contributions from William Ramsay, who discovered noble gases, further expanded the table. Collectively, these achievements have shaped modern chemical science and classification.
British contributions to the periodic table and chemistry
British scientists have made significant contributions to the periodic table and chemistry. John Dalton developed atomic theory, laying the groundwork for understanding chemical elements. Sir Humphry Davy discovered several elements, including sodium and potassium. Henry Moseley’s work on atomic numbers redefined the periodic table’s structure. Contributions from William Ramsay, who discovered noble gases, further expanded the table. Collectively, these achievements have shaped modern chemical science and classification.
💡 Key Takeaways
- Understand how John Dalton's atomic theory laid the groundwork for element classification and chemical predictions.
- Learn about Sir Humphry Davy's discovery of sodium and potassium and his impact on electrochemistry.
- Explain Henry Moseley's law and how it established atomic numbers as the organizing principle of the periodic table.
- Appreciate the lasting impact of British scientists on the development and modern structure of chemistry and the periodic table.
❓ Frequently Asked Questions
Who proposed the modern atomic theory and why is it important?
John Dalton proposed that matter is made of indivisible atoms; elements consist of atoms, and compounds form from whole-number ratios of atoms, laying the groundwork for modern chemistry.
How did Sir Humphry Davy contribute to element discovery?
Davy used electrolysis to break down compounds and isolate elements, notably sodium and potassium, showing elements could be produced from compounds by electricity.
What did Henry Moseley demonstrate about the periodic table's arrangement?
Moseley showed that atomic number, not atomic weight, determines an element's properties, leading to the modern periodic table arrangement and correcting previous ambiguities.
How do these British contributions shape our understanding of chemistry today?
They laid foundational theories, enabled discovery of new elements, and clarified how the periodic table is organized, guiding modern chemistry.
