Quantum Foundations & Atomic Orbitals
Quantum foundations refer to the fundamental principles and theories that explain the behavior of matter and energy at the smallest scales, such as atoms and subatomic particles. Atomic orbitals are regions around an atom’s nucleus where electrons are most likely to be found, as described by quantum mechanics. Together, these concepts help scientists understand the structure of atoms, chemical bonding, and the nature of matter at the quantum level.
Quantum Foundations & Atomic Orbitals
Quantum foundations refer to the fundamental principles and theories that explain the behavior of matter and energy at the smallest scales, such as atoms and subatomic particles. Atomic orbitals are regions around an atom’s nucleus where electrons are most likely to be found, as described by quantum mechanics. Together, these concepts help scientists understand the structure of atoms, chemical bonding, and the nature of matter at the quantum level.
💡 Key Takeaways
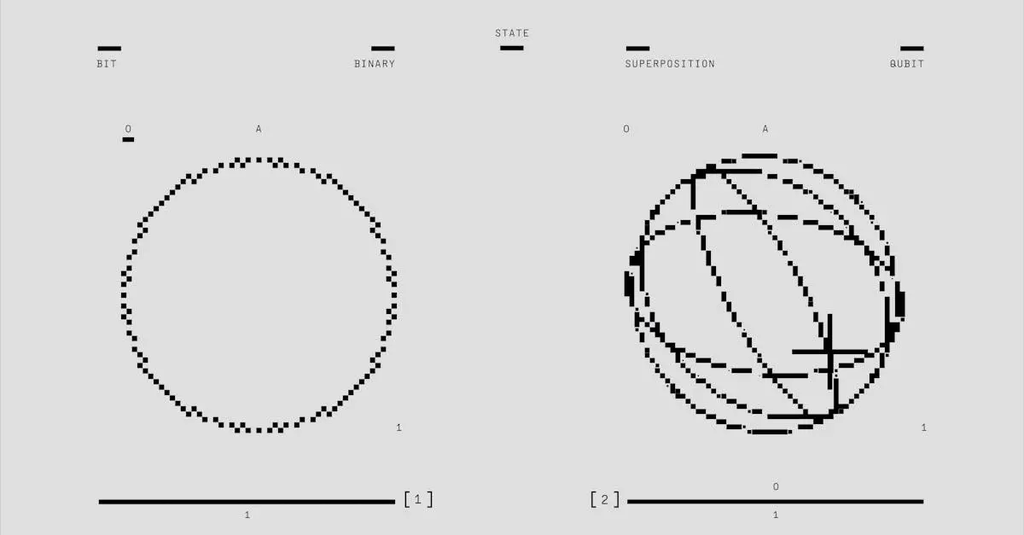
- Grasp the core ideas of quantum foundations: wave–particle duality, uncertainty, and probabilistic descriptions via the wavefunction.
- Understand atomic orbitals as probabilistic regions around the nucleus with shapes such as s, p, and d.
- Recognize energy quantization and orbital structure: electrons fill discrete shells and subshells (n, l, ml) leading to spectra.
- See how orbital properties influence chemistry: bonding, reactivity, and magnetic behavior.
❓ Frequently Asked Questions
What are quantum foundations?
The basic principles of quantum mechanics that describe how matter and energy behave at the smallest scales, including wave-particle duality, superposition, quantization, and probabilistic outcomes.
What is an atomic orbital?
A region in space where an electron is most likely to be found, described by a wave function with a specific energy and shape (s, p, d, f) rather than a fixed path.
How are orbitals related to electron shells?
Shells are energy levels (n). Orbitals are the spatial regions within those levels; each shell contains one or more orbitals, and each orbital holds up to two electrons with opposite spins.
Why do orbital shapes matter in chemistry?
Shapes like s, p, d, and f arise from solving the Schrödinger equation and influence how electrons participate in bonding and determine molecular geometry.
What does probability mean for electrons in atoms?
We describe electrons with probability densities given by the square of the wave function; we can predict where an electron is likely to be, not its exact position.
